-
The Vander Waals equation of state is {P + (a / c²)} (v – b)= RT, where p is pressure, v is specific volume, T is temperature and R is characteristic gas constant. The SI unit of a is
-
- J/kgK
- m3/kg
- m5/kg-s2
- Pa/kg
Correct Option: C
| p + | both term should gave same unit since they are getting added | v² |
| ⇒ | N | = a | 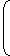 | kg |  | ² | ||
| m² | m3 |
| ⇒ a (unit) = | .kg. | kg² | s²m² |
| = | = | kg² s² | kgs² |

